Raising Larvae in the Zebrafish International Resource Center Autonursery
(Source: C. Barton from Zebrafish Book 5th Edition)
Introduction
The Zebrafish International Resource Center (ZIRC) nursery utilizes an autonursery system to raise larval fish. An autonursery system can significantly reduce labor in the nursery because hand siphoning and cage cleaning are eliminated. Fish can also be fed earlier without the concern of diminished water quality.
An autonursery system is any system that delivers water to a nursery tank and allows it to exit the tank via an appropriately sized mesh drain. There are commercial autonursery systems available from some fish rack system manufacturers. Because these commercial autonursery systems are usually made to work with a specific company's rack system, it is often not feasible for an existing facility to incorporate a production model system into their existing nursery. Many facilities have developed their own autonursery system using materials readily available to the research community and often at a fraction of the cost of a commercial unit.
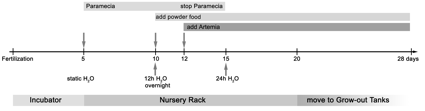
Before entering the autonursery at day 4, embryos are kept in Petri dishes at a density of 50 fish per dish. The Petri dishes are filled with 0.5xE2 embryo medium (Nüsslein-Volhard and Dahm, 2002). With 0.5xE2, the pH is more consistent and the osmolarity is between the range of standard embryo medium and our fish water. This rearing solution offers a more stable environment than standard fish water. Petri dishes are also dosed with methylene blue (dilution: 2ml of 0.1% methylene blue, to 1 liter of medium). This suppresses fungal outbreaks in Petri dishes, in the event that an egg goes bad. Petri dishes are also kept in a 29°C incubator.
There are three feeds used in the nursery: Paramecia, San Francisco Bay Brine Shrimp, and Zeigler AP Larval Diet. Detailed instructions on how to make each of these foods are covered in the Food Production and Recipes section.
Overview of autonursery protocol
Feeding
- 0-9 day old fish are fed concentrated paramecia
- 10-11 day old fish are fed concentrated paramecia and Zeigler larval diet
- 12-15 day old fish are fed concentrated paramecia, Zeigler, and brine shrimp
- 16-21 day old fish are fed brine shrimp and Zeigler
Water Changes
- 0-4 day old fish are kept in Petri dishes, in 0.5x E2 embryo media (See RECIPES, Chapter 10)
- 4 day old fish are moved into autonursery tanks, without any water flow
- 5-9 day old fish remain in the autonursery, no water flow
- 10-15 day old fish are given 12 hour water flow, only at night
- 15-21 day old fish are given 24 hour water flow
- 21+ day fish are transferred out of the nursery and into the grow-out section of the main facility.
Fig. 1. Overview of location, water and food changes in the ZIRC Autonursery during the first 4 weeks of development. Top, Water changes. Middle (light shading), Location/container changes. Bottom, Feeding schedule. During the first 4 days (shaded area), Embryos and larvae are kept in an incubator at 29 ºC, in 0.5x Embryo medium (Nüsslein-Volhard and Dahm) to provide stable environmental conditions for early development. |
Detailed procedures for feeding in the nursery
Day 0-4:
- Fish are kept in Petri dishes at a density of 50 fish per dish. They are also kept in a 0.5x E2 embryo media containing methylene blue to suppress any mold or fungal infections.
- During this time, the Petri dishes are kept in an incubator at 29°C. Only incubators with a glass front or programmable light cycles are appropriate for use in this application.
Day 4:
- Fish are transferred to an autonursery cage, with a small amount of fish water.
Day 5-9:
- During this time, fish are kept in autonursery cages with the water flow turned off.
- Fish are fed 40 ml concentrated paramecia (see recipe section at the end of this chapter), 2 times per day.
Day 10:
- On this day, a new food is added to the feeding regimen. In addition to the 40 ml concentrated paramecia, the babies are given Zeigler Larval Diet. Feed each cage 5 ml prepared Zeigler (see recipe section at the end of this chapter).
- Once Zeigler is added to the diet on day 10, the water to each cage is turned on for 12 hours during the night. The water flow should be no more than a slow drip. The water is turned off each morning before the feeding takes place.
Day 11:
- Fish are fed 40 ml concentrated paramecia and 5 ml Zeigler 2 times per day
- Water is running only during the night, and remains off during the day
Day 12:
- On this day, brine shrimp are added to the diet. Each cage is fed a 5ml portion of brine shrimp, 2 times per day.
- All other food and water conditions remain the same
Day 13-14:
- During these days, cages on the autonursery are fed 40 ml concentrated paramecia, 5 ml Zeigler, and 5 ml brine shrimp 2 times per day.
- Water is turned on only at night, and remains off during the day.
Day 15:
- On this day, Paramecia are removed from the babies diet. Zeigler and brine shrimp remain in the babies diet, and are fed 2 times per day.
- The water is changed to 24 hour flow.
Day 16-21:
- During these days, fish are fed brine shrimp and Zeigler 2 times per day.
- Water is on 24 hours a day.
Day 21:
- Fish are usually ready to be removed from the nursery and put into adult fish aquaria.
- If fish are not large enough, continue caring for them in the same manner until they are big enough to be transferred out of the nursery.
Day 21-80:
- Fish are put into a special nursery grow out space in the main facility. They are put into adult tanks, with special baby drains, which are perforated, but with small enough holes to retain the babies in the aquarium.
- They are fed brine shrimp and a baby powdered mix, consisting of 250-450 micron Zeigler powdered food (see recipe section at end of chapter).
- Once fish are large enough, approx. 2-3 months, their baby drains are replaced with adult drains and they are given adult master mix. They are also moved off the grow-out racks and onto the standard fish racks.
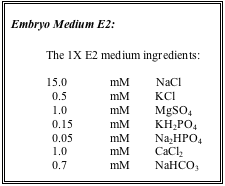
Use 0.5X E2 as a working solution to provide a smooth transition between the conductivity of embryo medium (~ ca. 1000 µS) and system water (around 600 µS).
Prepare three stock solutions, called E2A, E2B and E2C and use them to make a larger volume of the 0.5X E2.
E2A Stock: |
E2B: |
E2C: |
TO MAKE 20 LITERS 0.5X E2: |
Food Production and Feeding Zeigler powdered food recipe and feeding instructions
Ingredients: 150> Ziegler Larval Diet Powder150-250 Ziegler Larval Diet Powder
ZIRC Larval Diet Zeigler Mix: |
To prepare for nursery feeding, suspend a 0.5 gram spoonful of powder in 250 ml fish water. Feed each cage of 50 fish 5 ml.
Brine Shrimp
The Zebrafish International Resource Center recommends the use of San Francisco Bay brine shrimp cysts for larval zebrafish. They are 20-30% smaller on average than regular brine shrimp cysts, and therefore can be eaten by younger larval zebrafish. The adult fish are fed standard brine shrimp. If separate nursery cones are not available, standard brine shrimp can be substituted for San Francisco Bay brine shrimp.
There are two main methods for raising brine shrimp, decapsulated and non-decapsulated:
Standard (non-decapsulated) method
Ingredients:
• Dechlorinated tap water
• Rock salt
• Baking soda
• Brine shrimp cysts
Amounts of each ingredient depend on the amount of brine shrimp and the size of cone used. The following recipe is for a 16 liter cone and can be altered depending on your facility's needs:
• 16 liters dechlorinated tap water
• 400 ml Rock Salt
• 16 liters dechlorinated tap water
• 400 ml rock salt
• 1 1/2 tsp baking soda
• 50 ml _Artemia cysts
Other Supplies:
- Small air pump for each cone, or an air pump large enough to supply both cones
- Brine shrimp hatching cones, available from most aquaculture equipment suppliers
- 105 µm polyesterfilter cloth mesh basket (see Fig. 2 in the section on Paramecia)
Directions for setting up the cones:
Brine shrimp cones should be kept in a room with 24 hour light and a temperature of approximately 75-80ºF. Small cones can also be kept in light boxes if they are set up inside a fish room. The cone should be set up and cysts added approximately 18-24 hours prior to use. The late morning or early afternoon is usually the best time to do this so that the brine shrimp are ready for the first feeding by the next morning. Because of the time needed to hatch, two cones are used. This allows one cone to be hatching, while the other one is still being used to feed the fish throughout the day.
Set up the first cone with water, rock salt, baking soda, and brine shrimp cysts. The cones need to be aerated. To do this, connect a length of flexible air tubing to the pump outlet. To the end of the flexible tubing, connect a piece of rigid air tubing that is long enough to reach the bottom of the cone. Adjust the pump so that the cone is staying mixed, but not bubbling over. The brine shrimp will be ready for use in approximately 18-24 hours from the time the cysts were added, depending on factors such as light intensity and room temperature.
Collection and feeding with non-decapsulated brine shrimp:
Remove the air stick from the cone and allow it to settle for approximately 5 minutes. Once the shrimp have settled, use the spigot at the bottom of the cone to drain off 100 ml of brine shrimp. Using a 105 µm mesh basket and fish water, rinse the brine shrimp gently to remove all the salt. Pour the collected brine shrimp into a 1 liter squeeze bottle and fill with fish water. Each tank of 50 babies should receive a 1 second squirt of this dilution, 2 times per day.
Decapsulation Method
Decapsulation of brine shrimp is a procedure that removes the cyst shell before hatching. Cysts are soaked in a chemical treatment that removes the shell. The brine shrimp are then stored in a saturated salt solution and refrigerated. Cysts remain viable in this state for upwards of 6 weeks. There are several benefits of decapsulating brine shrimp prior to use:
- Decreased chance of fish ingesting cyst shells, which can impact the digestive system.
- Air does not have to be removed to collect brine shrimp for feedings. This decreases the number of brine shrimp that die and spoil in the cone during repeated collections. It also decreases the time required to prepare brine shrimp for daily feedings.
- More accurate feeding dilutions, because you can drain a consistent amount from the cone without having to remove the air.
To use the decapsulation method, cysts need to be prepared in advance before they can be hatched for food. The following procedure is a step by step guide to decapsulating brine shrimp cysts. It includes all the materials needed for the process.
Brine shrimp decapsulation procedure:
Materials:
- 15 oz can of dried Artemia cysts (approximately 430 g)
- 4.3 liters ~6% laundry grade bleach, chilled to 4ºC
- 1.25 kg rock salt (NaCl)
- 125 ml 40% lye (NaOH) solution (w/v)
- 30.0 g sodium thiosulfate (Na2S2O3)
- 16 liter hatching cone with aeration
- 125 µm mesh bag (Aquatic Eco-Systems PMB3, 125 µm x 18")
- Several 3-5 liter beakers
- 1-2 squirt bottles - squeeze type
Solutions:
*Should be prepared in advance kept chilled at 4ºC prior to use.
Bleach, ~6% Laundry Grade: |
Salt Solution 25 ppt: |
Lye (NaOH) Solution 40% (w/v): |
Buffered Salt Solution: |
Sodium Thiosulfate Solution 1.0%: |
Saturated Brine: |
Procedure:
1. Cyst hydration: Hydrate one full can of dried cysts in 5 liters of tap water in a hatching cone with aeration for 1 hour at room temperature. Examine the cyst under a stereoscope with top lighting before proceeding. Dry cysts are dimpled, resembling a deflated basketball, whereas fully hydrated cysts are completely spherical in shape. The cysts must be fully hydrated before decapsulation. If cysts are not completely spherical after 1 hour, continue the hydration process (for a maximum of 2 hours), checking the progress of the cysts under a microscope every 15 minutes.
2. Filter and rinse cysts: Collect the hydrated cyst in a 125 µm mesh bag and rinse with cool tap water.
3. Transfer cysts back to the cone with the chilled Buffered Salt Solution and aerate (save a filled squirt bottle of salt solution to help transfer cysts to cone).
4. Decapsulation: Add the chilled bleach (4.3 liters) to the cone and continue aeration. Watch the cysts turn from brown to grey to orange. When the cysts are 90% orange, stop the reaction by quickly siphoning the cysts through a 125 µm mesh bag and rinsing well with cool tap water.
5. Neutralization of residual chlorine: To neutralize any residual chlorine transfer the mesh bag to a clean 4 liter beaker and pour the 1.0% Sodium Thiosulfate (3 liters) into the bag. Soak the cysts in the Sodium Thiosulfate Solution for ~1 minute, then rinse the cysts with dechlorinated tap water.
6. Dehydration for long-term storage: Transfer the cysts back to the cone with 4 liters of saturated brine and aerate for 18-24 hours (save a filled squirt bottle of saturated brine solution to help transfer cysts to cone). Add granular NaCl as needed to keep the solution saturated during the dehydration process. Transfer dehydrated cysts to (5 or 6) 1 liter bottles and fill with clean saturated brine. Store in refrigerator.
Cone Set Up and Feeding Instructions
Cone set up and ingredients are the same for both decapsulated and non-decapsulated brine shrimp. The only difference is in how cysts are measured before they are added to the cone. Because the decapsulated cysts are stored in a liquid, the liquid must be removed to obtain an accurate volume.
Using the same idea as the paramecium filter baskets, make small sieves to remove the liquid and measure the cyst volume at the same time. The appropriate size mesh is affixed to the bottom of either a small plastic beaker or a large plastic culture tub. Cut off the bottom of the container so that it lines up with an existing measurement line in the beaker.
Before measuring the cysts, shake each bottle well. Then, pour it through the measuring sieve until the desired amount is reached, making sure that most of the water has been removed. The cysts can then be added to the cone.
Collection and feeding instructions using decapsulated brine shrimp
When using decapsulated brine shrimp, the shrimp do not need to settle in the cone before they are collected for feeding. Without removing the air line, measure 800 ml of liquid from the cone. As with the non-decapsulated method, rinse the brine shrimp using a 105 µm basket. Add the rinsed brine shrimp to a 1 liter bottle and fill with fish water. The feeding amount remains the same, 1 squirt per tank of 50 fish 2 times per day.
Additional reading
C. Nüsslein-Volhard (Ed.) and R. Dahm (2002) Zebrafish, Practical Approach, Oxford University press, Oxford UK