A High-Throughput Method For Sperm Cryopreservation And In Vitro Fertilization
(Source: B. Draper and C. Moens from Zebrafish Book 5th Edition)
This is a method for zebrafish sperm cryopreservation that is an adaptation of the Harvey method (Harvey et al., 1982). There are two primary changes to the original protocol that both streamline the procedure and increase sample uniformity. First, all sperm volumes are normalize using freezing medium that does not contain the cryoprotectant (i.e. methanol). Second, cryopreserved sperm are stored in cryovials instead of capillary tubes. The rates of sperm freezing and thawing (ƼC/time) are probably the two most critical variables to control in this procedure. For this reason, do not substitute different tubes for those specified. Working in teams of 2, it is possible to freeze the sperm of 100 males/team in ~2 hrs.
Sperm Freezing Solutions
Ginsberg Fish Ringers (make fresh every 3 days; Ginsburg, 1963):
NOTE: ORDER OF ADDITION IS IMPORTANT TO PREVENT PRECIPITATION
In 450 ml sterile ddH2O dissolve:
NaCl 3.25 g
KCl 0.125 g
CaCl2•2 H2O 0.175 g
Then add:
NaHCO3 0.10 g
Bring final volume to 500 ml with sterile ddH2O and store at 4ºC.
Freezing Medium (make fresh daily):
A. WITHOUT Methanol
Ginsburg Fish Ringers 10 ml (room temp.)
Powdered Skim Milk 1.5 gm
B. WITH Methanol
NOTE: ORDER OF ADDITION IS IMPORTANT TO PREVENT PRECIPITATION
Ginsburg Fish Ringers 9 ml (room temp.)
Methanol 1 ml
Powdered Skim Milk 1.5 g
After assembling freezing medium, mix well for 20 min on orbital shaker or rocker. Before using, aliquot into 1 ml Eppendorf tubes, avoiding surface bubbles.
Materials needed for high throughput sperm freezing
10 µl disposable pipettes (Fisherbrand cat# 22-358697)
250 ml beakers for fish water containing MESAB/Tricaine
watch glasses (Pyrex cat# 9985-75)
P20 Pipetter (or equivalent) and tips
Sponge fish holder (to hold male while squeezing)
Dissecting microscope with above stage lighting
Forceps (e.g. Millipore type)
2.0ml cryogenic vials (Corning cat# 430488)
10X10 cryoboxes (Nalgene cat#03-337-7AA)
Styrofoam container filled with ~ 15 cm of finely crushed dry ice
15 ml conical tubes (Falcon # 352099)
Dewar flask containing liquid nitrogen
Long tongs (e.g. CMS Fisher Health Care cat# 10-316B)
Cryogloves
Recovery tank for males
One Eppendorf tube filled with freezing media without methanol
One Eppendorf tube filled with freezing media with methanol
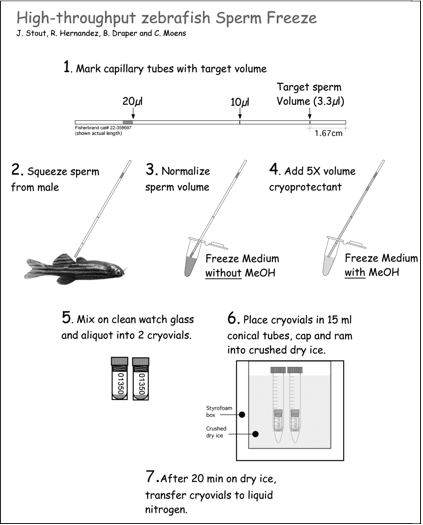
Figure 20. Sperm freezing procedure
Detailed Sperm Freezing Procedure
1. MARK CAPILLARY TUBES: Before starting, mark the 10 μl capillary tubes with a lab pen at the level of 3.33 µl (i.e. 16.7 mm from bottom; see Figure 20, above).
2. ANESTHETIZE MALE: Place male(s) in beaker containing MESAB/tricaine diluted in fish water (see RECIPES, chapter 10).
3. DRY FISH: Once anesthetized, gently blot fish dry on paper towel, paying special attention to dry the ventral side. Water activates sperm so it is important to dry around the cloaca thoroughly. Position fish on sponge holder, ventral side up (Fig. 20 panel 2). If the region around the anal fin is still moist, gently dry with a Kimwipe. Take care to not squeeze sperm prematurely!
4. Place marked capillary tube in rubber mouth pipette adapter that is included with the capillary tubes. The adaptor is a rubber hose that has a mouthpiece on one end and a capillary holder on the other.
5. Place male under the microscope and expose the urogenital opening by carefully spreading the anal fins apart using the end of the capillary tube.
6. COLLECT SPERM: Expel sperm by gently stroking the sides of the fish with smooth forceps (e.g. Millipore), or by gently squeezing the sides of the fish between your index finger and thumb. Collect sperm in capillary tube as it is expelled using gentle suction (Fig. 20 panel 2). Avoid feces that may be expelled with sperm.
7. NORMALIZE SPERM VOLUME TO 3.3 µl: If the volume of sperm reaches, or exceeds the pen mark on the capillary tube (i.e. 16.7 mm or greater), then proceed to step
8. If sperm volume does not reach target volume, then normalize the volume to pen mark using Freezing Medium WITHOUT Methanol (Fig. 20 panel 3). The minimum amount of sperm that is acceptable varies with quality of sperm. Good sperm is white and opaque. Poor sperm looks watery. In general, accept as minimums: 1 µl good sperm (or 1/3 target) and 2 µl poor sperm (or 2/3 target). Volume is now normalized to 3.3 µl.
8. ADD CRYOPROTECTANT TO SPERM: Suck up Freezing Medium WITH Methanol to the orange mark (approximate total volume is now 20 µl; Fig. 20 panel 4). Expel sperm and cryoprotectant mixture onto clean area of a watch glass, paying special attention not to introduce bubbles. Gently mix by pipetting up and down ~ 4 times (avoid introducing bubbles).
9. PLACE 10 μl SPERM INTO EACH OF 2 CRYOVIALS*: *Pipette 10 µl of the sperm + cryoprotectant solution into the bottom of two separate cryovials that have been labeled with relevant information (Fig. 20 panel 5). Cap vials and drop them into the bottom of room temperature 15 ml Falcon tubes- one cryovial/tube. Cap Falcon tube.
10. FREEZE SPERM FOR 20 MIN. ON DRY ICE: Immediately insert the pair of cryovial-containing Falcon tubes into crushed dry ice. The tubes should be inserted into the dry ice deep enough that only their caps show (Fig. 20 panel 6). To keep track of tubes in dry ice, number pairs of tubes from 1-20 (on caps) and record the time they go into the dry ice. A simple method for making finely crushed dry ice is to wrap it in a towel and pulverize it with a hammer.
11. PLACE CRYOVIALS INTO LIQUID NITROGEN: After the sperm samples have been frozen on dry ice for 20 min, transfer them to a liquid nitrogen-containing Dewar flask. Sperm is stored here until it is time to place the samples into the liquid nitrogen freezer. When placing in the freezer boxes, place freezer box in a bath of liquid nitrogen so that samples do not warm. Use long metal tongs to recover vials from the Dewar flask and handle with cryogloves.
12. Store cryovials long term in a cryogenic liquid nitrogen freezer. To maintain viability of sperm, it is important that vials are stored immersed in liquid nitrogen. Vials stored in the vapor phase lose viability over time.
Speed is important! The time between adding the cryoprotectant to placing sperm vials in dry ice (i.e. steps 6-10) should be no more than 30 sec.
In Vitro Fertilization Using Cryopreserved Sperm
This works best with two people. One person squeezes eggs from females while the other person thaws the sperm.
1. Set a water bath to 33ºC.
2. Remove cryovial from liquid nitrogen freezer and transfer to liquid nitrogen-containing Dewar flask until ready to thaw.
3. Squeeze eggs from females into 35 mm plastic Petri dish. If possible, try to obtain 3 clutches of eggs and combine into one dish. If you cannot get three good clutches within 1 min of your first clutch, then proceed to step 4 (one good clutch is sufficient). Definition of good clutch: >150 uniformly nice looking eggs (i.e. yellowish, with no white debris indicative of degradation). Keep dish covered while sperm is thawed.
4. Remove vial from liquid nitrogen and remove cap. Quickly immerse vial ~1/2 way into 33ºC water bath for 8-10 sec.
5. Quickly add 70 µl room temperature Hanks to vial and mix by pipetting up and down. Immediately add to eggs and mix gently with pipette tip.
6. Without delay, activate sperm and eggs by adding 750 µl fish water. Swirl to mix. Incubate 5 min at room temperature.
7. After 5 min, fill dish with fish water and incubate dish at 28ºC. After 2-3 hrs, count and transfer fertile embryos to 100 mm dishes (50/dish). Count infertile eggs so that fertilization frequency can be determined.
8. Take good care of the larvae! For the first 5 days, change water daily and remove dead embryos. Put only the larvae that have swim bladders into nursery on day 5.
References
Ginsburg, A. S. (1963) Sperm-egg association and its relationship to activation of the egg in salmonid fishes. J. Embryol. Exp. Morphol. 11: 13-33.
Harvey, B., R. N. Kelley and M. J. Ashwood-Smith (1982) Cryopreservation of zebra fish spermatozoa using methanol. Can. J. Zoology. 60: 1867-1870.